
Looking for How Does Home Food Preserving (Canning Fruits and Vegetables) Work? in 2024? Scroll down this page and follow the links. And if you bring home some fruit or vegetables and want to can, freeze, make jam, salsa or pickles, see this page for simple, reliable, illustrated canning, freezing or preserving directions. There are plenty of other related resources, click on the resources dropdown above. If you are having a hard time finding canning lids, I've used these, and they're a great price & ship in 2 days.
If you have questions or feedback, please let me know! There are affiliate links on this page. Read our disclosure policy to learn more.
How Does Home Food Preserving (Canning Fruits and Vegetables) Work?
How Does Canning Work, in a Nutshell?
Acidity, heat-treatment and a safe storage vessel are the basic keys to safe home canning.
Acidity
An acid food environment inhibits the growth of bacteria. The acidity of a food may occur naturally as in citrus fruits, apples, tomatoes and strawberries or it may be produced in foods through microbial fermentation. Selected acid producing bacterial cultures added directly to foods can produce desirable products like yogurt, buttermilk and fermented meat products. Acid may also be added directly to a food; an example is the addition of acetic acid to fish and vegetables, lactic acid to Spanish-type olives and citric acid to beverages.
The strength of acidity of a food is expressed by its pH value. The pH of a food is one of several important factors that determine the survival and growth of microorganisms during processing, storage and distribution. Consequently, food processors are interested in determining the pH of foods and in maintaining pH at certain levels to control microbial growth and prevent product deterioration and spoilage.
The pH scale was developed from mathematical calculations based on the dissociation temporary breakdown) of water. These complex calculations allow us to measure pH on a scale that runs from 0 to 14. The values that are less than 7 are acidic, while those greater than 7 are alkaline. A pH value of 7 is neither acid or alkaline and is considered neutral. Pure water has a pH of 7 and is neutral. The pH scale is based on the hydrogen ions concentration [H+] in the food. The more hydrogen ions present, the more acid the food and the lower the pH. The diagram below shows the pH scale and the taste of foods at acid and alkaline pH values.
| High-acid Foods | Acid Foods | Low-acid foods | Non-acid foods |
| 3.7 and lower | 3.7 to 4.5 | 4.6 to 5.3 | 5.4 and higher |
|
|
|
|
What does the acidity do?
In home canning foods, pH is used to control the growth of microorganisms by:
- directly inhibiting microbial growth, and
- reducing the heat resistance of the microbes.
Why does pH matter?
Every microorganism has a minimum, an optimum and a maximum pH for growth. Most microorganisms grow best at pH values around 7.0 while only a few grow below pH 4.0. Yeasts and molds are generally more acid tolerant than bacteria and can grow at lower pH values. Foods with pH values below 4.5 are usually not easily spoiled by bacteria but are more susceptible to spoilage by yeasts and molds. Microorganisms can grow in wide pH ranges and these ranges are probably the difference between different bacterial strains, types of food or growth medium and the type of acid or base used to adjust pH.
Click here for a listing of the approximate pH of foods and food products.
Next, heat treatment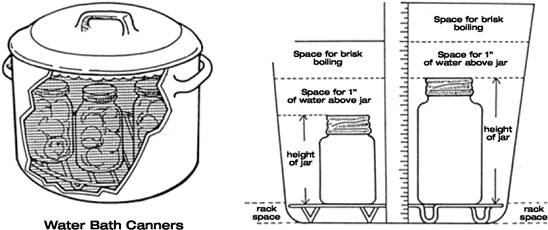
Heat is used to destroy active , living bacteria and many bacterial spores. Some spores still survive, but the acid environment will prevent them from becoming activing and growing.
Vegetables are predominately low-acid foods and require a severe heat process to destroy all spores of Clostridium botulinum . There are two ways to safely preserve these: some must be processed in a Pressure Canner that reaches higher temperatures (240 F) than a water bath canner, which is the other method.
Most fruits are naturally acid and may be given a mild heat process in which the temperature does not exceed 212°F and does not require pressure. This is called "water bath canning".
In both cases, food scientists test recipes in a lab, over and over, until the find the combination of ingredients, acidity, preparation (like chopping to a certain size), cooking and then processing (water bath or pressure and for how long), until they find a combination that rteduces bacteria to a low enough level to create safe storage and consumption of the sealed food.
For non-acidic foods that may be acidified like cucumbers, artichokes, cauliflower, peppers and fish, it is essential that the pH be allowed to equilibrate (stabilize) thoroughly before the heat treatment. This involves the addition of sufficient acid (typically either lemon juice or vinegar, as called for in the lab-tested recipe), proper mixing, and enough time for the pH to f all to 4.6 or below, in the center of solid foods. This MUST be determined in a lab; you cannot simply make up your own recipe by guessing how much vinegar or lemon juice to add!
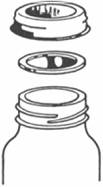
narrow metal screw band
metal lid with sealing compound
seals here
Sealing and storing the food in a safe vessel
Having followed a lab-tested recipe to create the proper acidic mixture, heating it to reduce active bacteria, we're down to the last step: sealing in a proper vessel and then a final heat treatment to ensure that jar and it's contents are "sanitized", which is like sterilizing... except not quite as thoruough. Home food preserving equiptment can't reach the temperatures need to destroy every single bacterial spore (the goal of sterilization), but it can reduce them to a level that prevents food borne illness (sanitization).
The hot food is ladeled into clean jars and the 2 peice lid and ring is screwed down snugly. Then the jars are loaded into the canner, the lid put on and it's heated to boiling for the length of time required by the tested recipe.
The genius of this system is that as the contents of each jar heats up, they expand and a bit of the air left in the top of the jar squeezes out by lifting the lid against the ring just enough to allow the air to escape, without letting water in.
Then when the jars are removed to cool down, the ring keeps the
lid in place, the contants shrink a bit and a vacuum is formed.
THat seals the lid downm, the glue in the lid keeps it sealed and
the ring is removed. Now you have a sanitized, sealed jar of
food good for room temperature storage for many months or even
years.
Looking for canning equipment and supplies?
Water bath canner with a jar rack
Pressure canners for gas, electric and induction stoves: Presto 23Qt or T-fal 22Qt
Canning scoop (this one is PERFECT)
Ball Blue book (most recent version)
Jars: 8oz canning jars for jams
Find Other types of farms:
Farm markets and roadside stands
Road trips and camping resources
Local Honey, apiaries, beekeepers
Consumer fraud and scams information
Home canning supplies at the best prices on the internet!
Maple Syrup Farms, sugarworks, maple syrup festivals
Environmental information and resources
Farms For Your Event for birthday parties, weddings, receptions, business meetings, retreats, etc.
Festivals - local fruit and vegetable festivals
Get the
most recent version of
the Ball Blue Book
With this Presto 23 quart pressure canner and pressure cooker, you can "can" everything, fruits, vegetables, jams, jellies, salsa, applesauce, pickles, even meats, soups, stews. Model 01781

You can make jams, jellies, can fruit, applesauce, salsa and pickles with water bath canners, like this Granite Ware 12-Piece Canner Kit, Jar Rack, Blancher, Colander and 5 piece Canning Tool Set

