
Looking for Oregon Cottage Food Labels - How to Label Homemade Foods in Oregon in 2024? Scroll down this page and follow the links. And if you bring home some fruit or vegetables and want to can, freeze, make jam, salsa or pickles, see this page for simple, reliable, illustrated canning, freezing or preserving directions. There are plenty of other related resources, click on the resources dropdown above. If you are having a hard time finding canning lids, I've used these, and they're a great price & ship in 2 days.
If you have questions or feedback, please let me know! There are affiliate links on this page. Read our disclosure policy to learn more.
Oregon Cottage Food Labels - How to Label Homemade Foods in Oregon
Oregon Cottage Food Labeling
If you fall under the Oregon cottage food laws (which is primarily applicable to home-baked breads, cakes, pies, cookies, doughnuts, crackers and hard candies (see complete list here) , then you still must label the food products according to the Oregon requirements. Here are examples, details and label templates to use.
 Labeling requirements
Labeling requirements
Cottage Food Production Operations must label all of their food products properly, which include the following information on the label of each unit of food product offered or distributed for sale:
- The name of the product,
- net weight,
- ingredient statement, and
- the name and address of the producer.
If the item is perishable, an expiration date is required.
It is a good idea to submit this label to the Food Safety Program for review before having a quantity printed. There is a handout available that further explains the labeling requirements (electronically available on the website).
Allergen labeling
The federal Food Allergen Labeling and Consumer Protection Act of 2004 (FALCPA) requires that foods containing any of the eight major food allergens are clearly labeled on the principal display panel of the food. The eight major allergens are:
- Milk (any protein from milk, butter, cream, dry milk, whey, or casein)
- Eggs (e.g., whites, yolks, albumen, or powdered eggs)
- Soy (e.g., soy beans soy lecithin, soy protein, soy, or soy flour)
- Wheat (includes spelt, semolina, kamut, and triticale)
- Seafood (e.g., salmon, tuna, eel, bass, flounder, or cod)
- Crustacean shellfish (e.g., crab, lobster, or shrimp)
- Peanuts (e.g., peanut butter or peanut meal)
- Tree nuts (e.g., pecans, walnuts, hazelnuts, cashews, coconut, or pine nuts)
Labeling Options
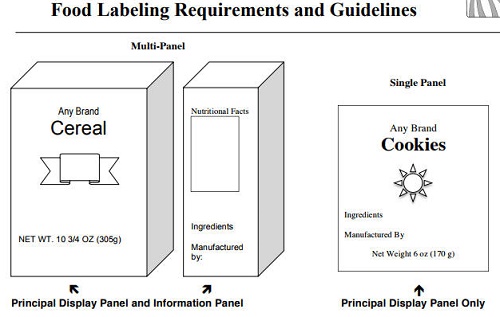
There are two labeling options as shown in the diagram above.
- All required information may be captured on the principal display or
- may be spread out between the Principal Display Panel and the Information Panel.
Firms that use a stamp or affix a sticker to label their product will usually choose to include the information on the Principal Display Panel only.
All labels shall be in English, with exemptions for other languages as discussed in 21CFR 101.15.
Principal Display Panel
The Principal Display Panel (PDP) is the portion of the package that is most likely to be seen by the consumer at the point of purchase. Although a brand name may appear on the principal display panel, this is voluntary. The PDP must include the product identity and net weight, but may also include the firm's address, nutritional facts and the ingredients if a single panel is used.
Product Identity
The truthful or common name of the product that is contained in the package. This is located in the middle portion of the PDP front label. The product identity must be of dominant and of contrasting type. Artwork or company's logos must not hide or detract from the prominence of the product identity. Minimum type height is 1/16" of a lower case "o".
Net Weight
The net weight must be located in the lower 30% of the PDP. Net weight must appear as a distinct item, separated from other print. The net weight or volume must be in both the US lb/oz (Avoirdupois oz.) and metric scale. "Net Wt. 12 oz (340 g)" for a dry product and "Net 32 fl. oz (1QT) 946 ml" for a liquid product. Minimum type height is 1/16th". In some instances (e.g. pickles and some bakery items) may be sold by count instead of weight. The net weight must be listed as the minimum net weight, not by the average net weight. Note: Net weight is the gross weight minus the tare weight of the packaging. The net weight declaration print size is specific to the size of the area on the principal display panel (21 CFR 101.105(h)(3)(i)(1-4)).
If the principle display panel is:
Less than 5 sq. inches ---------------------1/16" high
5 to 25 sq. inches --------------------- 1/8" high
25 to 100 sq. inches ---------------------3/16" high
100 to 400 sq. inches ---------------------1/4" high
Information Panel

This is the area normally to the right of the principal display panel. On a box, it would be the label on the right side of the package; on a jar it would be directly to the right of the principal display panel. The information panel must contain certain information in the order specified below.
Nutrition Facts
All packaged foods require a nutrition fact statement panel, unless they qualify for an exemption in 21 CFR 101.9 (h-j). Additionally, some small firms may qualify for an exemption of the nutritional facts statement; http://1.usa.gov/1J32fs3 If a nutrition or health claim is made on a packaged food, a nutrition facts panel is required.
Ingredient Statement
This statement is generally located below the nutrition facts and above the manufacturer or distributor information. The ingredients including food colorings and chemical preservatives must be listed in descending order of predominance by weight or volume. If less than 2% by weight, the ingredient can be listed at the end with a statement "contains 2% or less of ___". Without exception, allergens must always be declared regardless of weight, volume or percentage. The ingredient shall be the common or usual name of the ingredient except that certain spices and flavorings need to be declared according to regulation (21CFR 101.22). Spices, such as paprika, turmeric, saffron and others that are also colorings must be declared either by the term 'spice and coloring' or by the actual (common or usual) names, such as 'paprika' (21 CFR 101.22(a)(2)). An ingredient which itself contains two or more ingredients and which has an established common or usual name must be followed by a parenthetical listing of all ingredients contained therein in descending order of predominance.
Place of Address
The place of business is located on the lower portion of the label. The name, street address, city, state and zip code of the food manufacturer or the distributor must be listed. However, the street address may be omitted if it is shown in a current city directory or telephone directory. A telephone number or a website may be listed but is not a requirement. If a person manufactures, packs, or distributes a food at a place other than his principal place of business, the label may state the principal place of business in lieu of the actual place where such food was manufactured or packed or is to be distributed, unless such statement would be misleading. When the person whose name appears on the label does not manufacture the packaged food, the name shall reflect the connection of the person with an appropriate term such as "manufactured by" or "distributed by".
Additional Requirements
Many manufactured foods may have further requirements, for example:
- Bottled water has additional labeling and nomenclature requirements about the source (21CFR 165.110)
- Perishable foods are required to have a pull date (OAR 603-025-0800) and should be labeled "Keep Refrigerated".
- SAFE HANDLING INSTRUCTIONS are required on certain types of foods (i.e., eggs, juice) (21CFR 101.17) and meats.
- Packaged foods designated for export must comply with the foreign
countries labeling requirements.

Due to the complexity of your product, there may be other labeling requirements and options specific to your product. These include the use of colors, flavorings, when to declare water as an ingredient, and the use of the term 'fresh' . Dietary ingredients include vitamins, minerals, amino acids, and herbs or botanicals, as well as other substances that can be used to supplement the diet. These items have special labeling requirements, and for an overview the following website may be helpful: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm050803.htm
Sample Labels and Templates
Here is a free Microsoft Word label template which you can download and edit. These labels are already formatted to fit on Avery Template 22820 Print-to-the-Edge Oval, Labels 2" x 3-1/3", 8 per Sheet, Glossy White. You can get the label stock online (see at right).
Questions? Contact Information:
If you have questions or need more information, you may contact the Food Safety Program office in Salem at 503-986-4720.
Food Safety
635 Capitol St NE
Salem, OR 97301
Phone:
503-986-4720
Looking for canning equipment and supplies?
Water bath canner with a jar rack
Pressure canners for gas, electric and induction stoves: Presto 23Qt or T-fal 22Qt
Canning scoop (this one is PERFECT)
Ball Blue book (most recent version)
Find Other types of farms:
- Easter egg hunts
- Children's consignment sales
- Farm markets and roadside stands
- Road trips and camping resources
- Local Honey, apiaries, beekeepers
- Local Meat, Milk and Eggs
- Consumer fraud and scams information
- Home canning supplies at the best prices on the internet!
- Maple Syrup Farms, sugarworks, maple syrup festivals
- Environmental information and resources
- Farms For Your Event for birthday parties, weddings, receptions, business meetings, retreats, etc.
- Festivals - local fruit and vegetable festivals
- Pumpkin patches and corn mazes
- Christmas Tree Farms and lots
Get the
most recent version of
the Ball Blue Book
Get the
most recent version of
the Ball Blue Book of Home Canning
Find other types of farms:
- Easter egg hunts
- Children's consignment sales
- Farm markets and roadside stands
- Local Honey
- Local Meat, Milk and Eggs
- Road trip and camping
- Pumpkin patches and corn mazes
- Christmas Tree Farms and lots
- Maple Syrup farms and sugarworks
Highly rated canning supplies:
- Regular Mouth Canning Lids with food-grade with BPA Free Silicone seals for Ball, Kerr Jars for Canning
- Canning accessories kit: funnel, jar tongs, lid lifter, etc
- Food Dehydrator, 400W Electric with 8 Trays, 48 hour Timer and Temperature Control 95-176℉, BPA-Free
- Water Bath Canner, 21 Qts with lid, Jar Rack, Speckled Black, cans 7 quart jars, 9 pint jars or 13 half-pint jars
- Air Fryer: Instant Pot Instant Vortex Plus XL 8QT Clear Windows, Custom Programming, 8-in-1 Functions that Crisps, Broils, Roasts, Dehydrates, Bakes, Reheats
- Pressure canner: All American 921, 21.5qt Pressure Cooker/Canner, never needs gaskets, Great for Gas, Electric or Flat Top Stoves - Made in the USA
- The Backyard Homestead:a guide to homesteading , on 1/4 acre, how to raise grains and vegetables; raise animals for meat, eggs, and dairy; and keep honey bees
- Smart silent HEPA Air Purifiers for Home, Large Rooms for Allergies, Smoke, Pets. Eliminates 99.97% of Dust, Pet Hair, Odors


